Such molecules have minimum solubility in water or salt solutions at the pH that corresponds. Place the amino acids that possess a charge on their side chain at physiologic pH into the bin. 
The side chains of some amino acids are charged at physiologic pH. This procedure can of course be extended to the amino acids with acidic side chains (aspartic acid glutamic acid) and those with basic side chains (lysine arginine histidine). The pI value can affect the solubility of a molecule at a given pH. The appropriate pKa values are 3.9 and 9.7, which average to 6.8 for the -1 charged state. You'll find that since the side chain has a lower $\mathrm pK_\mathrm a$ than the amino group, you average the carboxyl and the side chain $\mathrm pK_\mathrm a$'s. The same logic applies to cysteine ( look up the $\mathrm pK_\mathrm a$ values and draw out the differently protonated forms). interactions (orbital overlap) between the pi bonds of aromatic rings. If the side chain $\mathrm pK_\mathrm a$ were lower than $9.11$, then you should average the carboxyl and side chain $\mathrm pK_\mathrm a$'s instead. The author tells us AMC is liberated from a peptide as a result of peptide bond. pKa1 is the -carboxyl group, pKa2 is the -ammonium ion, pKa3 is the side chain group if applicable and pI is the isoelectric point at which the amino acid. Our peptide calculator is a convenient tool for scientists as a molecular weight peptide calculator, which can be used as an amino acid calculator as well. 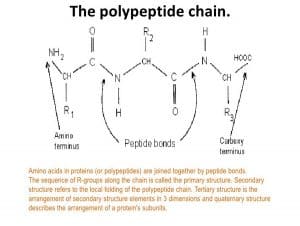
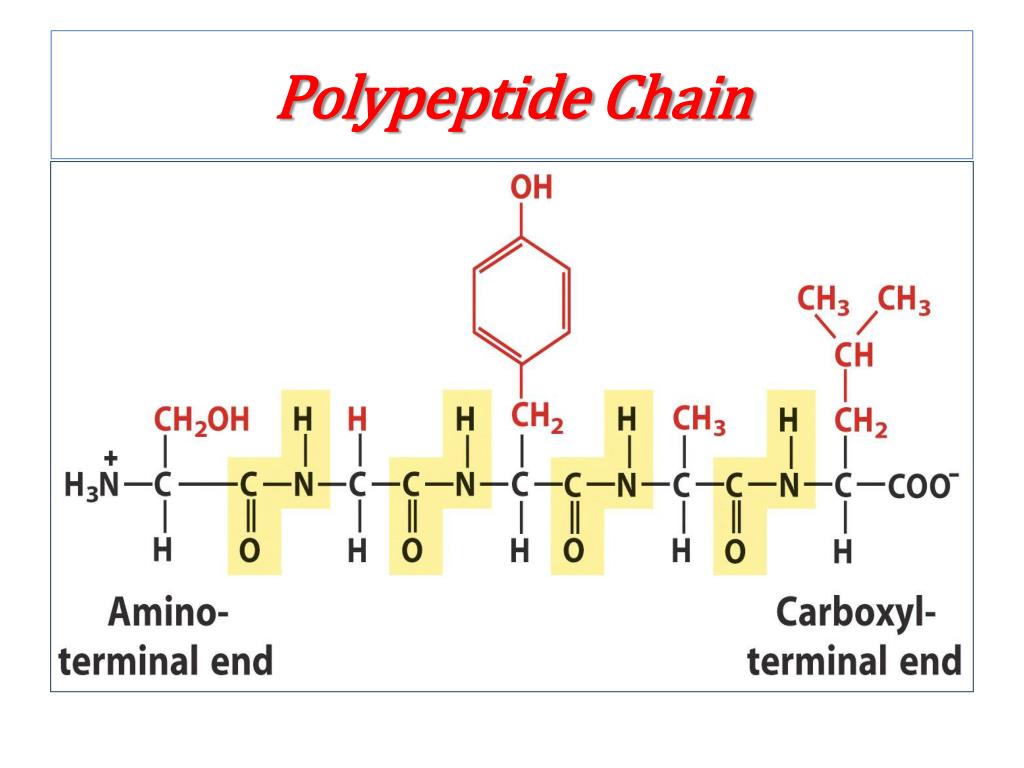
The peptide bond can only be broken by hydrolysis, where the. The ammonium holds the proton more tightly than does the acid. It just so happens that $2.20$ is the carboxyl $\mathrm pK_\mathrm a$ and $9.11$ is the amino $\mathrm pK_\mathrm a$. To quickly calculate the isoelectric point (pI) of a peptide you need to follow three steps: 1. Long chain polypeptides can be formed by linking many amino acids to each other via peptide bonds. The pKa of the acid is near 5, and the pKa of the ammonium is near 9. Since the $\mathrm$ of tyrosine is $5.66$ (the average of $2.20$ and $9.11$). 
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
